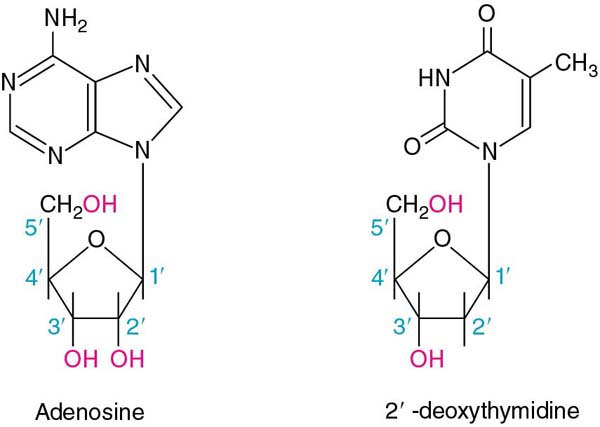
Now we join one of the purines or pyrimidines to the sugar (an N-glycosidic bond to the 1' carbon of the sugar). These structures are called nucleosides:

Notice that the numbering convention for the sugar is now 1' through 5'.
When phosphates are now added to the 5' carbon, we have the final structures to consider, the nucleotides:
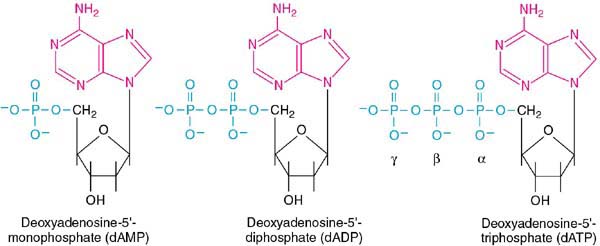
We now add the final bit of nomenclature convention. The phosphates are designated, moving out from the 5' carbon, as alpha, beta, and gamma. This is very important to remember, since the position of radiolabeled phosphorous in experimental protocols will determine the outcome.
The nomenclature of the nucleosides found in RNA and DNA are:
|
Base |
Nucleoside |
Deoxynucleoside |
| adenine | adenosine | deoxyadenosine |
| guanine | guanosine | deoxyguanosine |
| cytosine | cytidine | deoxycytidine |
| uracil | uridine | (not found in DNA) |
| thymidine | (not found in RNA) | (deoxy)thymidine* |
Note: For thymidine the deoxy is understood. In fact in the rare occurrence of T in RNA (in some transfer RNAs) the nucleoside is called ribothymidine.
The nomenclature of the nucleotides use the names of the nucleosides plus the number of phosphates (see the figure above).